Research overview
I am retired. These are the broad research areas in which I have been involved.
The Early Years - 1975 - 1990
ECD (Electronic Circular Dichroism)
I have always been interested in chirality and chiroptical properties. Being computer chemists, mr group undertook to develop a program to calculated optical rotatory strengths from ab initio wavefunctions. The challenge here was to obtain a description of an excited state wavefunction that was comparable to the ground state post-Hartree Fock wavefunction. A brilliant student, Jose Barriel (1950 - 1976), succeeded spectacularly..
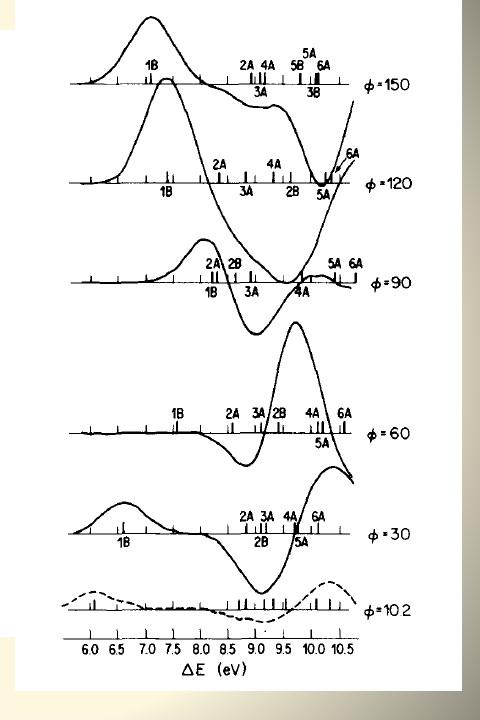
More recently - 1988 - 1998
VCD (Vibrational Circular Dichroism)
Under the impetus of Hal Wieser (1935 - 2020), who had built the first instrument for measuring vibrational circular dichroism spectra in Canada, my group undertook to study the phenomenon theoretically. Two brilliant students, Remo Dutler and Danya Yang, implemented the Vibronic Coupling Theory of Nafie and Freedman at the ab initio level to interface with Gaussian.
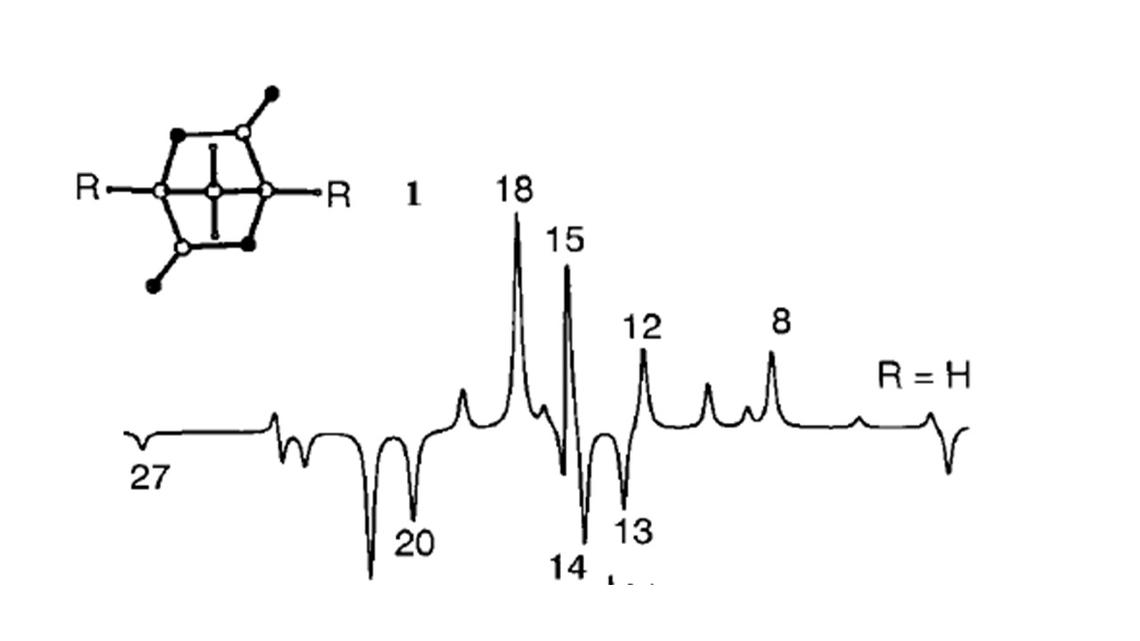
Career-long Interests
Properties of Reactive Intermediates
Carbocations
Carbocations are organic compounds with an electron deficient tricoordinated carbon atom. For many years, I had a fruitful interaction with the late Ted Sorensen (1934 - 2017) who studied carbocations, species whose structures are non-classical and highly fluxional, undergoing interesting rearrangements governed by principles of orbital interactions. I taught an course, Advanced Organic Chemistry, based on explaining structure and reactivity by the principles of orbital interactions, and was incouraged byTed and an organic colleague and friend, Mike Benn () to write up the notes in the form of a book which was published by Wiley-Interscience, in 1994 (1st edition), and 2000 (2nd edition)
Organic Free Radicals
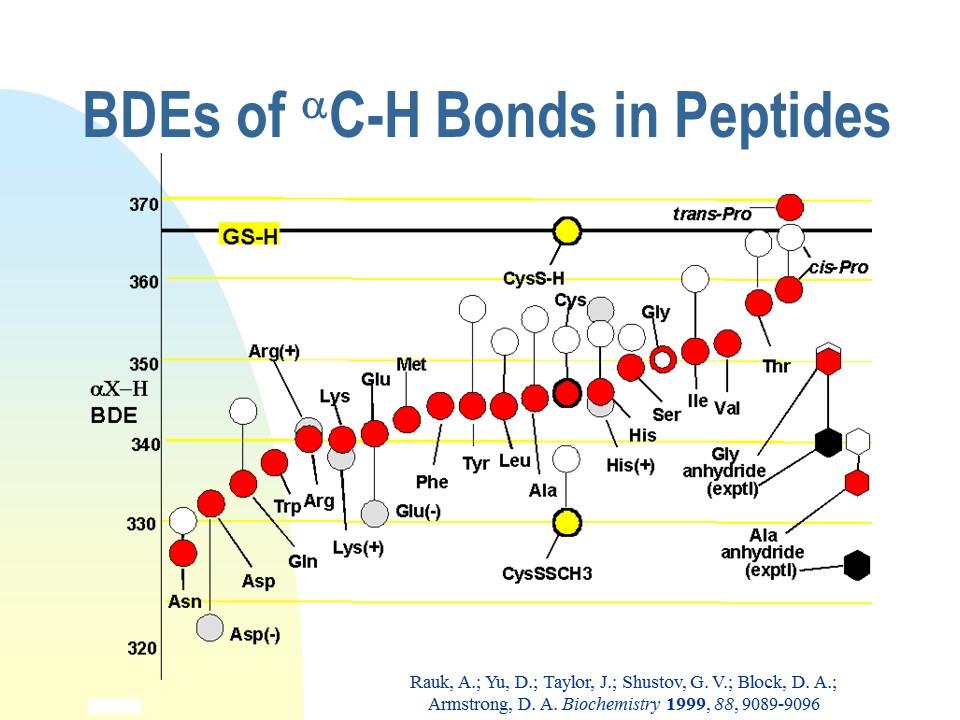
Organic Free radicals are organic compounds with a tricoordinated carbon atom bearing a single electron. In collaboration with Eugene Tschuikow-Roux (1936 - 2016) we examined the structures and stabilities of simple alkyl C-centred free radicals. With David Armstrong (1930 - 2006) we investigated the structures and stabilities of C-centred radicals bearing electron donor and electron acceptor groups, both of which are predicted to be stabilized by orbital interaction theory. Exceptionally stable are those with both donating and accepting substituents (captodative stabilization).

Published by John Wiley & Sons, 2000
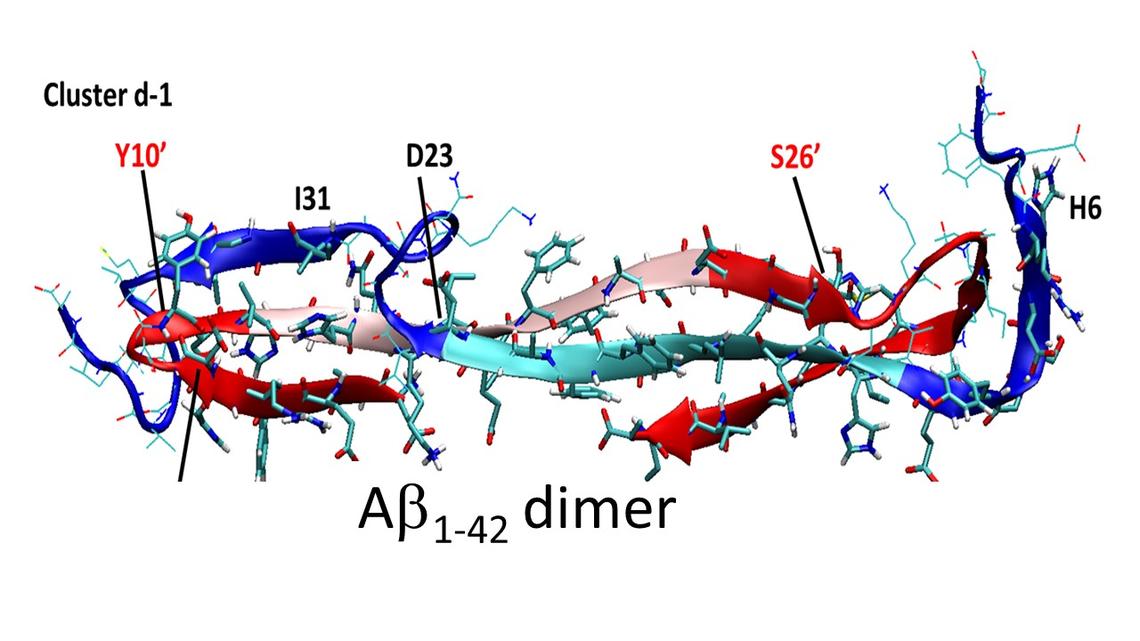
Retirement Years - 2000 - 2020
The Chemistry of Alzheimer's Disease
Following the realization that a C-centred radical at the aC site of every amino acid is exceptionally stable by the captodative effect, we examined oxidative damage to proteins and were led immediately to the amyloid beta (Ab) peptide of Alzheimer's disease. What followed was a 20 year investigation of the chemistry of Ab and a search for compounds that might inhibit its toxicity.